
ARC-Grain Crops, Potchefstroom

Dr Belinda Janse
van Rensburg,
ARC-Grain Crops, Potchefstroom
Diplodia ear rot occurs sporadically over seasons depending on climatic conditions. In epidemic seasons (early season dry weather and excessive wet, warm weather during grain fill) it is possibly one of the most damaging ear rot diseases in Southern Africa.
This ear rot is caused by two fungi in the same genus namely, Stenocarpella maydis and S. macrospora (previously named Diplodia maydis and D. macrospora which inspired the common name Diplodia ear and stalk rot). Stenocarpella macrospora has a polycyclic life cycle (the fungus can produce spores from leaf lesions throughout the season). The monocyclic S. maydis produces a sudden flush of spores simultaneously being released after a rain spell that followed a long, dry period. Stenocarpella macrospora ear rot symptoms are almost identical to that of S. maydis ear rot, but each occurs under different climatic and epidemiological conditions.
They must be treated as two separate diseases which vary in terms of their epidemiology even though they visually appear to be identical. The reason is that control measures for S. maydis ear rot are not always directly applicable to S. macrospora ear rot. If a producer is not certain which ear rot organism is present, it is necessary to contact a local plant pathologist. These two pathogens can only be correctly identified by a pathologist upon isolation onto artificial media and then identified based on spore size and shape. In this article the authors will focus on Diplodia ear and stalk rot caused by S. maydis.
Stenocarpella macrospora ear rot generally appears to be predominant in areas of KwaZulu-Natal, but was recently also isolated from an infected ear from the Makwassie district, suggesting a possible shift in the spread of the two pathogens within the South African maize industry. Under favourable conditions (seasons with early drought followed by excessive and extended rainfall during the maturation stage of the maize plant), S. maydis ear rot can occur throughout the entire maize production area of South Africa. Diplodia ear rots result in yield loss (when light-weight rotten kernels are discarded in the harvesting process), grain grade reductions and toxicity problems for animals. Effects of these mycotoxins on humans have not yet been adequately studied.
Diplodia stalk rot (Photo 1) is common in seasons where early season rainfall is followed by a midseason drought period during grain fill. These conditions cause unexpected stress on the maize plants at this critical period, which predisposes the plants to increased withdrawal of nutrients from the stems that leads to an increase in their susceptibility to various stalk rot pathogens. Diplodia stalk rots may also occur when the roots and crown are infected by fungi such as Gibberella root and crown rot. The infected plant is prevented from taking up nutrients and water during grain fill, which again stresses the plant when it is now obliged to use nutrients from the stem to ensure grain fill. This forced withdrawal of sugars and nutrients from the stalk predisposes the stalk to infection by stalk rot pathogens such as Diplodia stalk rot. Leaf diseases reduce the photosynthetic ability of the plant leaves and predisposes the plant to Diplodia and other stalk rots.

Symptoms
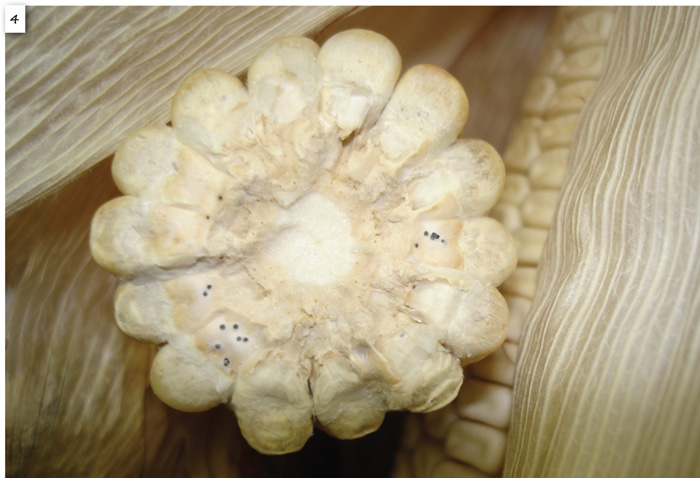
Diplodia ear rot symptoms associated with early infections during early ear development include yellowing and drying of husk leaves while stalks and leaves remain green (Photo 2). Infection generally starts at the ear base and ramifies upwards in the ear. The entire ear becomes overgrown with a white mycelial growth (Photo 3). External symptoms of infections that occur during grain fill is difficult to see unless the ear is opened by pulling back the ear husk leaves. A cross section of a mature infected ear shows black spore-producing bodies (pycnidia) at the kernel bases (Photo 4). Late season infections may occur when kernel moistures are already low while plants are retained in the field to dry down. These symptoms are less obvious. Kernel embryos become infected and slightly discoloured but no fungal ramification on the rest of the ears is observed. Such symptomless infections are locally referred to as ‘skelm Diplodia’.


Generally Diplodia stalk rot symptoms are noticed for the first time when maize plants start lodging (Photo 5). Infections in the stem occur and gradually result in the disintegration of the integrity of the stalk. This disintegration can only be observed once stalks are cut open and the disintegration of the stalk pith tissue is observed. Unlike most other stalk rots which discolour when infected, Diplodia-infected stalks remain white.

Economic importance
Yield losses caused by Diplodia ear rot are difficult to quantify due to various methods of harvest. For example, the make of harvester, harvesting speed and settings will all affect the percentage rotten kernels that remain in the grain bin or trailer. The percentage of lighter, rotten kernels that are blown out during the harvesting process depend on abovementioned factors. This implies that where Diplodia ear rot infections are present, the damage is aggravated in many cases. Firstly, if a low percentage of rotten kernels are discarded during the harvesting process, serious grain quality reductions occur. Secondly, where a high percentage of rotten kernels are discarded in the harvest process, it will improve grain quality but manifest itself as yield loss.
Diplodia stalk rot reduces the ability of the maize plant to reach its full yield potential by preventing adequate grain fill (reduced 1 000 kernel weight) and by preventing the pollinated ear from filling all the kernels on the ear to its full potential. This results in incomplete ears without grain developing on the cob towards the tip and kernels that are not packed as tight rows as on a healthy ear.
Mycotoxins, which have been shown to affect animals differently in various studies, are produced by Diplodia ear and stalk rot disease. Many of these mycotoxins were originally determined and reported in South Africa where field symptoms of diplodiosis were initially reported. Diplodiosis is defined as a nervous disorder of cattle and sheep resulting from the ingestion of mouldy cobs or stalks on a field infected by S. maydis. Reports have indicated that Diplodia-infected maize used in the chicken broiler and egg laying industries have resulted in reduced performance. Some S. maydis isolates were acutely toxic to ducklings and rats but were unable to induce diplodiosis in either cattle or sheep. This may indicate that more than one mycotoxin may be produced by the S. maydis isolates and that each fungal isolate may vary in their ability to produce certain mycotoxins. The wide range of toxicity and symptoms observed in animals may also be caused by specific mycotoxin combinations. The effect of these Diplodia-related mycotoxins on humans has not yet been determined. Continued research on Diplodia ear rot, stalk rot and their mycotoxins will improve our understanding of this complex disease and its mycotoxicology on a range of animals and possibly humans.
Incidence and severity
The incidence and severity of Diplodia ear and stalk rots are largely seasonal because climatic factors play a major role in the epidemiology of these two diseases. Epidemics of Diplodia ear rot have been associated with early season droughts and late season rains, particularly where infected stubble has been retained on the soil surface and susceptible hybrids have been planted. The epidemic of the mid 1980s in South Africa has since been followed by smaller, more localised occurrences. In these areas susceptible hybrids have been planted in a monoculture system where surface stubble retention was high from previous, yet often unnoticed Diplodia stalk rot epidemics. Diplodia stalk rot epidemics often go unnoticed because breeding programmes have focused on resistance to lodging (rind strength), which does not equate to resistance to the stalk rot pathogens. Plants may therefore still be infected by the stalk rot pathogens but do not lodge as easily.
Life cycle and epidemiology
Stenocarpella maydis spores are transmitted by air, seed and soil. Airborne spores cause heavy infections for up to 10 m from the inoculum source, but the number of successful infections is reduced with an increase in distance from the source. Single spores travelling long distances may lead to trace infections which may then develop into an epidemic focal point. Infected maize seed is an important inoculum source that leads to seedling and crown rot diseases, but the majority of S. maydis-infected kernels do not germinate.
Spores that land behind leaf or ear sheaths germinate and infect stalks or ears. Stalk infections may occur through root and crown infections where viable spores in the ground infect under favourable conditions and the fungus ramifies up through the crown into the lower internodes when the maize plant becomes stressed. Spore germination requires free water, but is inhibited by exposure to sunlight and desiccation. Infection may occur up to seven days after germination.
Germinated spores may enter tissue and lie dormant until conditions (generally related to physiological changes in the ear and stalk later in the season) are favourable for fungal growth through the tissues. Mycelial colonisation is accompanied by cell wall degradation ahead of the growing pathogen due to enzymes that are secreted by the fungus. Fungal ramification of maize ears starts at the shank, while cob tissue colonisation begins at the attachment with embryonic tissues and proceeds into the endosperm. Similarly stalk ramification may also occur. Infected tissues develop fruiting bodies (pycnidia) which produce spores during the following season.
Control measures for Diplodia ear and stalk rots
Stubble reduction/retention
Diplodia ear rot and Diplodia stalk rot involve the same pathogen and host, but should be seen as two separate diseases since climatic conditions influence the epidemics of each. As S. maydis predominantly survives on maize stubble rather than in the soil, any management practice that reduces levels of infected surface stubble will reduce inoculum concentrations in the field. Control of Diplodia ear rot includes surface stubble reduction through grazing, burning, baling or ploughing in of surface maize stubble. The removal of stubble for a single season only reduces Diplodia ear rot for that specific season. Where stubble is present the following season, the risk of Diplodia ear rot will increase to its original level under favourable weather conditions.
On the other hand, regenerative agriculture tillage practices improve soil water retention which, in turn, reduces plant stress levels and consequently, the plant’s predisposition to Diplodia stalk rot. Therefore, stubble retention may on the one hand increase Diplodia ear rot should the pathogen’s inoculum from the previous season survive on this stubble, but possible stalk rot incidence may be reduced which, in turn, may reduce inoculum levels for the following season. To reduce the possible effects of this contradiction, it is critical that Diplodia ear and stalk rot control be seen holistically and that other control measures be included in an integrated control programme to manage both Diplodia ear and stalk rots.
It is important to bear in mind that there are many advantages to using minimum or no-till systems and these are not to be ignored. Using alternate integrated control measures may enable a producer to retain the advantages of the conservation tillage practices and still reduce the incidence of Diplodia ear and stalk rot, particularly in seasons less favourable for disease development or under irrigation.
Stress reduction
Avoid planting unrealistically high plant populations on marginal soils and in areas where there is a high probability of drought, leaf or alternate stalk rot diseases. Ensure adequate and balanced plant nutrition relative to the yield potential of the land or area to be planted. Unnecessary stressors on the crop may increase the potential for Diplodia stalk rot which, indirectly in the long term, may increase inoculum levels on the land. Under conditions favourable for Diplodia ear rots an epidemic may occur, but no direct relationship between plant stress and Diplodia ear rot has been reported to date.
Crop rotation
Crop rotation reduces Diplodia ear rots by reducing inoculum levels in two ways. Firstly, a non-host for the fungus will not allow the fungus to persist for the season where maize is not grown. Secondly, a greater period (a season or two) between maize crops allows for natural breakdown of maize stubble, which further reduces survival of the fungus. Leguminous crops such as soybeans, dry beans, groundnuts and cowpeas are very good rotation crops. Wheat and oats can also reduce Diplodia ear rots. It is yet unclear why sunflower do not significantly reduce Diplodia ear rots under experimental conditions.
Early harvesting
Early harvesting will reduce Diplodia ear rots because the time available for the fungus to grow on the ear is reduced. Stenocarpella maydis can grow on maize ears in the field provided that their moisture level is 11% or more. Late or winter rains keep ears wet and increase the chance for fungal growth. In certain cases, it would be beneficial to harvest early at higher moisture levels and artificially dry the grain. (This practice is possibly why Diplodia ear rot is not considered a major problem in the United States.)
Hybrid resistance
The selection of cultivars is very important in the control of Diplodia ear and stalk rots. Four concepts involved in reactions of hybrids to diseases are tolerance, resistance, susceptibility and immunity. These all refer to different reactions of a hybrid to a specific disease. If a hybrid cannot get the disease, that hybrid is immune. If a hybrid is resistant, it does not imply that it will not get a specific disease, but resistance should be seen as a scale that runs from resistant to susceptible, with varying degrees of resistance/susceptibility in between. Resistance of one hybrid is also relative to that of other hybrids. For example, where the mean of all hybrids tested is 30% for Diplodia ear rot, any hybrid with less than that percentage is resistant and those above are considered susceptible. This mean percentage varies from one season to another or over localities within a season, depending on weather conditions and inoculum levels.
During a season where the mean ear rot of all hybrids is 5%, the resistant hybrid should still be resistant and the susceptible still be susceptible where hybrid reactions are consistent, because this effect is an interaction between the available inoculum, the host and prevailing weather conditions. A resistant hybrid will always have less Diplodia ear rot than susceptible hybrids relative to other hybrids and to prevailing climatic conditions. The local South African national cultivar trial entries were responsible for screening maize hybrids for resistance/susceptibility to Diplodia ear rots annually under various weather and inoculum conditions. Unfortunately, this project done by the ARC-Grain Crops (a neutral research institution) has been discontinued and seed companies are responsible for screening their own hybrids.
 Resistance to Diplodia stalk rot is difficult to quantify as plant standability or resistance to lodging because a thicker rind may mask Diplodia stalk rot infection. The pith within this thicker rind may, however, still be infected by Diplodia stalk rot and will break down. Thicker rinds have a resistance to decomposition, thereby increasing survival of S. maydis inoculum within intact maize residues, particularly those retained on the soil surface.
Resistance to Diplodia stalk rot is difficult to quantify as plant standability or resistance to lodging because a thicker rind may mask Diplodia stalk rot infection. The pith within this thicker rind may, however, still be infected by Diplodia stalk rot and will break down. Thicker rinds have a resistance to decomposition, thereby increasing survival of S. maydis inoculum within intact maize residues, particularly those retained on the soil surface.
Should an outbreak of lodging or Diplodia stalk rot occur, please contact the authors of this paper (018 299 6100) for scientifically based advice on the development of an integrated control system to reduce Diplodia ear rot or stalk rot to economically viable limits. The authors are also currently involved in research related to the mycotoxicology of this pathogen and need a wide range of isolates to be collected from different areas for further studies. In the same process the producer will have the causal organism correctly identified and get advice on controlling the diseases.




























